Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Report 2026

Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Report 2026
Global Outlook – By Test Type (In Vitro Renal Toxicity Testing, In Vivo Renal Toxicity Testing, In Silico Renal Toxicity Modeling), By Method (Biochemical and Enzymatic Assays, Cell Based Renal Assays, Biomarker Based Toxicity Analysis, Histopathology and Imaging, Pharmacokinetic and Drug Metabolism Studies), By Stage of Drug Development (Discovery and Lead Optimization, Preclinical Development, Clinical Development, Post Marketing Safety Studies),By End User (Pharmaceutical and Biotechnology Companies, Contract Research Organizations, Academic and Research Institutes, Regulatory Laboratories) – Market Size, Trends, Strategies, and Forecast to 2035
Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Overview
• Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing market size has reached to $1.52 billion in 2025 • Expected to grow to $2.78 billion in 2030 at a compound annual growth rate (CAGR) of 12.9% • Growth Driver: Increasing Kidney Disease Prevalence Boosts The Market Due To Rising Diabetes And Drug Safety Needs • Market Trend: Advanced Lab-On-A-Chip Solutions For Accurate Kidney Toxicity Assessment • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market?
Renal toxicity–absorption, distribution, metabolism, and excretion (ADME) toxicology testing is a specialized drug safety and pharmacokinetic evaluation approach that assesses the potential of pharmaceutical and biotechnology compounds to cause kidney damage by analyzing their absorption, distribution, metabolism, and excretion characteristics. This testing integrates in vitro, in vivo, and in silico methods to determine how drug candidates interact with renal tissues and to predict nephrotoxic risks during preclinical and clinical development. The main types of renal toxicity – absorption, distribution, metabolism, and excretion (ADME) toxicology testing include in vitro renal toxicity testing, in vivo renal toxicity testing, and in silico renal toxicity modeling. In vitro renal toxicity testing refers to laboratory-based methods that evaluate the potential toxic effects of compounds on kidney cells or tissues. These tests are conducted using methods such as biochemical and enzymatic assays, cell-based renal assays, biomarker-based toxicity analysis, histopathology and imaging, and pharmacokinetic and drug metabolism studies. They are applied across stages of drug development including discovery and lead optimization, preclinical development, clinical development, and post-marketing safety studies. They utilize technologies including high-throughput screening platforms, organ-on-chip and microfluidic systems, omics-based toxicology platforms, artificial intelligence and predictive modeling tools, and digital pathology and imaging systems, and are used by end users including pharmaceutical and biotechnology companies, contract research organizations (CROs), academic and research institutes, and regulatory laboratories.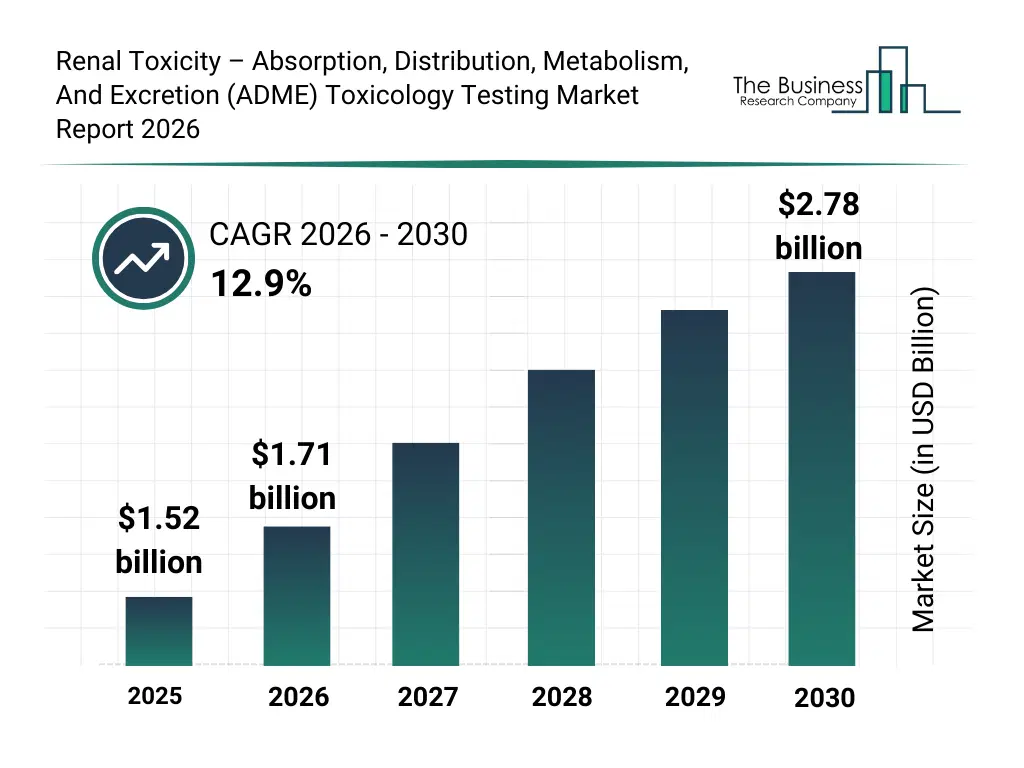
What Is The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Size and Share 2026?
The renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market size has grown rapidly in recent years. It will grow from $1.52 billion in 2025 to $1.71 billion in 2026 at a compound annual growth rate (CAGR) of 12.7%. The growth in the historic period can be attributed to growth in preclinical drug safety testing, rise in nephrotoxicity related drug failures, expansion of CRO services, increased regulatory safety requirements, growth in in vitro assay adoption.What Is The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Growth Forecast?
The renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market size is expected to see rapid growth in the next few years. It will grow to $2.78 billion in 2030 at a compound annual growth rate (CAGR) of 12.9%. The growth in the forecast period can be attributed to increase in biologics and complex drugs, predictive toxicology model adoption, expansion of organ on chip platforms, precision medicine research growth, AI based safety analytics demand. Major trends in the forecast period include kidney organoid based toxicity assays, AI assisted nephrotoxicity prediction models, high throughput renal screening platforms, biomarker driven renal safety panels, microphysiological kidney on chip systems.Global Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Segmentation
1) By Test Type: In Vitro Renal Toxicity Testing; In Vivo Renal Toxicity Testing; In Silico Renal Toxicity Modeling 2) By Method: Biochemical and Enzymatic Assays; Cell Based Renal Assays; Biomarker Based Toxicity Analysis; Histopathology and Imaging; Pharmacokinetic and Drug Metabolism Studies 3) By Stage of Drug Development: Discovery and Lead Optimization; Preclinical Development; Clinical Development; Post Marketing Safety Studies 4) By Technology: High Throughput Screening Platforms; Organ On Chip and Microfluidic Systems; Omics Based Toxicology Platforms; Artificial Intelligence and Predictive Modeling Tools; Digital Pathology and Imaging Systems 5) By End User: Pharmaceutical and Biotechnology Companies; Contract Research Organizations; Academic and Research Institutes; Regulatory Laboratories Subsegments: 1) By In Vitro Renal Toxicity Testing: Primary Renal Cell Assays; Immortalized Renal Cell Line Assays; Three Dimensional Renal Cell Cultures; Kidney Organoid Models; Transporter Interaction Assays; Biomarker Based Nephrotoxicity Assays; High Content Imaging Assays; Microphysiological Kidney System Models 2) By In Vivo Renal Toxicity Testing: Acute Renal Toxicity Studies; Subacute Renal Toxicity Studies; Subchronic Renal Toxicity Studies; Chronic Renal Toxicity Studies; Renal Biomarker Evaluation Studies; Histopathological Kidney Assessment; Dose Response Toxicity Studies; Recovery and Reversibility Studies 3) By In Silico Renal Toxicity Modeling: Quantitative Structure Activity Relationship Modeling; Machine Learning Based Toxicity Prediction; Physiologically Based Pharmacokinetic Modeling; Virtual Screening for Nephrotoxicity; Read Across Toxicity Modeling; Systems Biology Based Toxicity ModelingWhat Is The Driver Of The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market?
The rising prevalence of kidney-related disorders is expected to propel the growth of the renal toxicity-absorption, distribution, metabolism, and excretion (ADME) toxicology testing market going forward. Kidney-related disorders are conditions that damage the kidneys and affect their ability to filter blood and maintain body balance. Kidney-related disorders are increasing due to rising diabetes, which damages kidney blood vessels and impairs their filtering function. Renal toxicity ADME/toxicology testing helps detect and evaluate potential kidney damage caused by drugs or chemicals, enabling early intervention and safer treatment development. For instance, in 2025, according to the American Cancer Society, a US-based professional organization, approximately 80,980 new cases of kidney (renal) cancer are expected to be diagnosed in the US, with an estimated 14,510 deaths resulting from the disease. Therefore, the rising prevalence of kidney-related disorders is driving the growth of the renal toxicity-absorption, distribution, metabolism, and excretion (ADME) toxicology testing market.Key Players In The Global Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market
Major companies operating in the renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market are Thermo Fisher Scientific Inc., Eurofins Scientific SE, Agilent Technologies Inc., WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., PerkinElmer Inc., Beckman Coulter Inc., Bio-Rad Laboratories Inc., Pharmaron Beijing Co. Ltd., Simulations Plus Inc., GVK Biosciences Pvt. Ltd., Promega Corporation, Syngene International Ltd., SAI Life Sciences Ltd., QPS Holdings LLC, Southern Research Institute, BioIVT LLC, TCG Lifesciences Pvt. Ltd., Frontage Laboratories Inc., Symeres, TSRL Inc., BOC Sciences LLC, Vivisciences Preclinical CRO.Global Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Trends and Insights
Major companies operating in the renal toxicity-absorption, distribution, metabolism, and excretion (ADME) toxicology testing market are focusing on developing innovative solutions, such as low-drug-absorbing microfluidic chips, to improve predictive accuracy of kidney toxicity while reducing reliance on animal testing. A low-drug-absorbing microfluidic chip refers to a lab-on-a-chip device designed for renal toxicity testing that minimizes the unintended absorption of drugs by the chip material, ensuring more accurate simulation of kidney functions and reliable measurement of how drugs affect renal cells. For instance, in September 2024, Emulate Inc., a US-based biotechnology company, launched the chip-R1 rigid chip to enhance kidney toxicity testing with more accurate and reliable results. It is built for accurate ADME and toxicology modeling in non-stretch organs like the liver and kidney, using low-drug-absorbing plastics and a thin, porous polycarbonate membrane to prevent compound loss and boost drug recovery for lipophilic compounds. Its advantages include high shear stress support (up to 2.3 dyn/cm²), preactivated surfaces for easy workflows, and better cellular crosstalk, while benefits encompass improved reproducibility, precise toxicity detection, and compatibility with existing organ-chip hardware to accelerate human-relevant preclinical testing.What Are Latest Mergers And Acquisitions In The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market?
In April 2025, CN Bio Innovations Ltd., a UK-based organ-on-a-chip technology company, partnered with Pharmaron Beijing Co. Ltd. to integrate its organ-on-a-chip platform for drug testing and development. This partnership aims to integrate PhysioMimix organ-on-a-chip technology across Pharmaron's global R&D sites for toxicity testing, including renal applications, ADME studies, disease modeling, and co-developing innovative human-relevant models to enhance drug discovery while reducing animal testing. Pharmaron Beijing Co. Ltd. is a China-based company that offers renal toxicity ADME toxicology testing.Regional Insights
North America was the largest region in the renal toxicity-ADME toxicology testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market?
The renal toxicity–absorption, distribution, metabolism, and excretion (ADME) toxicology testing market includes revenues earned by entities through laboratory-based testing services, preclinical and clinical evaluation, data analysis, and regulatory support. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Report 2026?
The renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.71 billion |
| Revenue Forecast In 2035 | $2.78 billion |
| Growth Rate | CAGR of 12.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Method, Stage of Drug Development, Technology, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Eurofins Scientific SE, Agilent Technologies Inc., WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., PerkinElmer Inc., Beckman Coulter Inc., Bio-Rad Laboratories Inc., Pharmaron Beijing Co. Ltd., Simulations Plus Inc., GVK Biosciences Pvt. Ltd., Promega Corporation, Syngene International Ltd., SAI Life Sciences Ltd., QPS Holdings LLC, Southern Research Institute, BioIVT LLC, TCG Lifesciences Pvt. Ltd., Frontage Laboratories Inc., Symeres, TSRL Inc., BOC Sciences LLC, Vivisciences Preclinical CRO. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing market was valued at $1.52 billion in 2025, increased to $1.71 billion in 2026, and is projected to reach $2.78 billion by 2030.
request a sample hereThe global Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing market is expected to grow at a CAGR of 12.9% from 2026 to 2035 to reach $2.78 billion by 2035.
request a sample hereSome Key Players in the Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing market Include, Thermo Fisher Scientific Inc., Eurofins Scientific SE, Agilent Technologies Inc., WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., PerkinElmer Inc., Beckman Coulter Inc., Bio-Rad Laboratories Inc., Pharmaron Beijing Co. Ltd., Simulations Plus Inc., GVK Biosciences Pvt. Ltd., Promega Corporation, Syngene International Ltd., SAI Life Sciences Ltd., QPS Holdings LLC, Southern Research Institute, BioIVT LLC, TCG Lifesciences Pvt. Ltd., Frontage Laboratories Inc., Symeres, TSRL Inc., BOC Sciences LLC, Vivisciences Preclinical CRO. .
request a sample hereMajor trend in this market includes: Advanced Lab-On-A-Chip Solutions For Accurate Kidney Toxicity Assessment. For further insights on this market.
request a sample hereNorth America was the largest region in the renal toxicity-ADME toxicology testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here