Release Testing For Gene Therapy Market Report 2026

Release Testing For Gene Therapy Market Report 2026
Global Outlook – By Test Type (Potency Testing, Purity Testing, Identity Testing, Safety Testing, Sterility Testing, Other Test Types), By Product Type (Assay Kits, Reagents, Instruments, Services), By Therapy Type (In Vivo Gene Therapy, Ex Vivo Gene Therapy), By Application (Clinical Diagnostics, Research, Quality Control, Process Development, Other Applications), By End-User (Pharmaceutical and Biotechnology Companies, Academic and Research Institutes, Contract Research Organizations, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Release Testing For Gene Therapy Market Overview
• Release Testing For Gene Therapy market size has reached to $1.43 billion in 2025 • Expected to grow to $3.08 billion in 2030 at a compound annual growth rate (CAGR) of 16.5% • Growth Driver: Increasing Global Approvals of Gene Therapies Fueling The Growth Of The Market Due To Regulatory Progress And Expanded Therapeutic Pipelines • Market Trend: Advancing High-precision Digital Pcr Platforms For Gene Therapy Quality And Release Testing • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Release Testing For Gene Therapy Market?
Release testing for gene therapy is a quality control process conducted to ensure that gene therapy products meet predefined safety, potency, purity, and identity standards before they are administered to patients. It helps to confirm that gene therapy products are safe, effective, and compliant with regulatory requirements for clinical or commercial use. The main types of release testing for gene therapy include potency testing, purity testing, identity testing, safety testing, sterility testing, and other test types. Potency testing refers to procedures that assess the biological activity of gene therapy products to ensure efficacy and consistency. These tests are applied for product types including assay kits, reagents, instruments, and services. They are used for therapy types such as in vivo gene therapy and ex vivo gene therapy. They are applied across applications including clinical diagnostics, research, quality control, process development, and other applications, and are utilized by end users including pharmaceutical and biotechnology companies, academic and research institutes, contract research organizations (CROs), and other end users.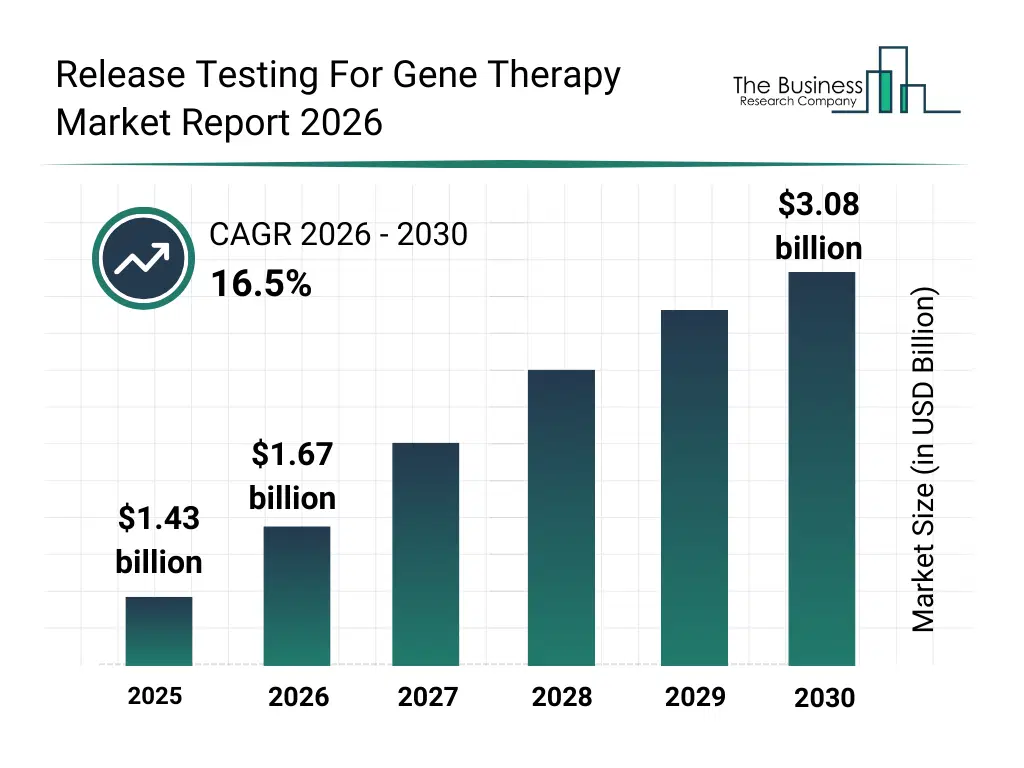
What Is The Release Testing For Gene Therapy Market Size and Share 2026?
The release testing for gene therapy market size has grown rapidly in recent years. It will grow from $1.43 billion in 2025 to $1.67 billion in 2026 at a compound annual growth rate (CAGR) of 16.3%. The growth in the historic period can be attributed to rise in gene therapy trials, strict regulatory quality standards, growth in viral vector production, expansion of Biologics Testing labs, increase in advanced therapy approvals.What Is The Release Testing For Gene Therapy Market Growth Forecast?
The release testing for gene therapy market size is expected to see rapid growth in the next few years. It will grow to $3.08 billion in 2030 at a compound annual growth rate (CAGR) of 16.5%. The growth in the forecast period can be attributed to growth in commercial gene therapies, rising demand for rapid release testing, expansion of contract testing services, increasing regulatory scrutiny, adoption of automated qc platforms. Major trends in the forecast period include high sensitivity potency assays, rapid sterility testing platforms, automated batch release analytics, multi parameter quality control panels, advanced vector identity testing.Global Release Testing For Gene Therapy Market Segmentation
1) By Test Type: Potency Testing; Purity Testing; Identity Testing; Safety Testing; Sterility Testing; Other Test Types 2) By Product Type: Assay Kits; Reagents; Instruments; Services 3) By Therapy Type: In Vivo Gene Therapy; Ex Vivo Gene Therapy 4) By Application: Clinical Diagnostics; Research; Quality Control; Process Development; Other Applications 5) By End-User: Pharmaceutical and Biotechnology Companies; Academic and Research Institutes; Contract Research Organizations; Other End-Users Subsegments: 1) By Potency Testing: Functional Activity Assays; Gene Expression Level Measurement; Biological Activity Evaluation; Dose Response Assessment 2) By Purity Testing: Residual Host Cell Protein Analysis; Residual Deoxyribonucleic Acid Quantification; Process Related Impurity Testing; Product Related Impurity Assessment 3) By Identity Testing: Genetic Sequence Confirmation; Vector Identity Verification; Transgene Identity Analysis; Cell Line Authentication 4) By Safety Testing: Adventitious Agent Testing; Replication Competent Virus Detection; Endotoxin Level Evaluation; Toxicity Assessment 5) By Sterility Testing: Bacterial Contamination Testing; Fungal Contamination Testing; Mycoplasma Detection; Environmental Monitoring Validation 6) By Other Test Types: Stability Testing; Packaging Integrity Testing; Shelf Life Determination; Transport and Storage Condition TestingWhat Is The Driver Of The Release Testing For Gene Therapy Market?
The increasing global approvals of gene therapies are expected to drive the growth of the release testing for gene therapy market going forward. Gene therapies are medical treatments that involve the introduction, removal, or modification of genetic material within a patient’s cells to treat or prevent disease. The growth in global approvals of gene therapies is supported by increasing regulatory acceptance, scientific advancements, and rising demand for innovative treatments for previously untreatable genetic disorders. Release testing for gene therapy is a regulatory requirement that ensures product quality and patient safety through potency, purity, identity, and safety assessments prior to batch release. For instance, in November 2023, according to the National Center for Biotechnology Information, a U.S.-based governmental research resource, more than 100 gene, cell, and ribonucleic acid (RNA) therapies had been approved globally as of early 2023, with over 3,700 additional therapies in clinical and preclinical development. Therefore, the expanding number of global gene therapy approvals is expected to contribute to the growth of the release testing for gene therapy industry.Key Players In The Global Release Testing For Gene Therapy Market
Major companies operating in the release testing for gene therapy market are Thermo Fisher Scientific Inc., Boehringer Ingelheim BioXcellence GmbH, Fujifilm Diosynth Biotechnologies Inc., AGC Biologics Inc., SGS SA, Lonza Group AG, Eurofins Scientific SE, WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., Sartorius AG, Samsung Biologics Co. Ltd., Bio-Rad Laboratories Inc., Pace Analytical Services LLC, KBI Biopharma Inc., BioAgilytix Labs LLC, Viralgen Vector S.L., GenScript ProBio LLC, KCAS Bioanalytical Services, Creative Biolabs LLC, Cellipont Bioservices Inc.Global Release Testing For Gene Therapy Market Trends and Insights
Major companies operating in the release testing for gene therapy market are focusing on developing innovative solutions, such as digital PCR-based analytical testing platforms, to address the growing need for highly accurate, sensitive, and standardized methods that ensure product safety, potency, and regulatory compliance for complex gene therapy products. Digital polymerase chain reaction (PCR)-based analytical testing platforms are advanced molecular systems that precisely quantify deoxyribonucleic acid (DNA) or ribonucleic acid (RNA) by partitioning samples into thousands of individual reactions, enabling highly sensitive and absolute measurement of nucleic acid targets. For instance, in April 2025, QIAGEN N.V., a Germany-based provider of molecular diagnostics and life science tools, expanded its QIAcuity digital PCR portfolio with new lentivirus solutions designed to enhance quality control in cell and gene therapy manufacturing. These additions support viral vector genome titration, vector copy number determination, and replication-competent lentivirus detection, enabling high-precision analytics for process development, batch release, and regulatory compliance. The platform offers absolute quantification across singleplex and multiplex applications, strong repeatability, and tailored kits for critical gene therapy vectors, providing more reliable and reproducible results than many conventional testing approaches.What Are Latest Mergers And Acquisitions In The Release Testing For Gene Therapy Market?
In January 2026, bioMérieux S.A., a France-based biotechnology company, acquired Accellix Inc. for approximately $41 million (€35 million). With this acquisition, bioMérieux aimed to strengthen and expand its pharmaceutical quality control portfolio particularly in rapid, automated flow cytometry solutions for cell and gene therapy manufacturing to support the growing advanced therapies market and improve testing capabilities across the therapy development lifecycle. Accellix Inc. is a US-based quality control technology provider that enables rapid, automated flow cytometry–based release testing for cell and gene therapies, specifically supporting critical release parameters such as cell purity and viability.Regional Insights
North America was the largest region in the release testing for gene therapy in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Release Testing For Gene Therapy Market?
The release testing for gene therapy market includes revenues earned by entities through the quality control and analytical testing services, stability assessment, method validation, regulatory compliance testing, and batch release support. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Release Testing For Gene Therapy Market Report 2026?
The release testing for gene therapy market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the release testing for gene therapy industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Release Testing For Gene Therapy Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.67 billion |
| Revenue Forecast In 2035 | $3.08 billion |
| Growth Rate | CAGR of 16.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Product Type, Therapy Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Boehringer Ingelheim BioXcellence GmbH, Fujifilm Diosynth Biotechnologies Inc., AGC Biologics Inc., SGS SA, Lonza Group AG, Eurofins Scientific SE, WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., Sartorius AG, Samsung Biologics Co. Ltd., Bio-Rad Laboratories Inc., Pace Analytical Services LLC, KBI Biopharma Inc., BioAgilytix Labs LLC, Viralgen Vector S.L., GenScript ProBio LLC, KCAS Bioanalytical Services, Creative Biolabs LLC, Cellipont Bioservices Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Release Testing For Gene Therapy market was valued at $1.43 billion in 2025, increased to $1.67 billion in 2026, and is projected to reach $3.08 billion by 2030.
request a sample hereThe global Release Testing For Gene Therapy market is expected to grow at a CAGR of 16.5% from 2026 to 2035 to reach $3.08 billion by 2035.
request a sample hereSome Key Players in the Release Testing For Gene Therapy market Include, Thermo Fisher Scientific Inc., Boehringer Ingelheim BioXcellence GmbH, Fujifilm Diosynth Biotechnologies Inc., AGC Biologics Inc., SGS SA, Lonza Group AG, Eurofins Scientific SE, WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., Sartorius AG, Samsung Biologics Co. Ltd., Bio-Rad Laboratories Inc., Pace Analytical Services LLC, KBI Biopharma Inc., BioAgilytix Labs LLC, Viralgen Vector S.L., GenScript ProBio LLC, KCAS Bioanalytical Services, Creative Biolabs LLC, Cellipont Bioservices Inc. .
request a sample hereMajor trend in this market includes: Advancing High-precision Digital Pcr Platforms For Gene Therapy Quality And Release Testing. For further insights on this market.
request a sample hereNorth America was the largest region in the release testing for gene therapy in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the release testing for gene therapy market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here