Pharmaceutical Validation Services Market Report 2026

Pharmaceutical Validation Services Market Report 2026
Global Outlook – By Service Type (Installation Qualification, Operational Qualification, Performance Qualification, Revalidation), By Product Type (Active Pharmaceutical Ingredient Validation, Finished Pharmaceutical Product Validation, Biologics and Biosimilars Validation, Medical Device Validation), By Delivery Mode (Onsite Validation Services, Remote Validation Services), By End-User (Pharmaceutical Manufacturers, Biopharmaceutical Companies, Contract Development and Manufacturing Organizations, Contract Research Organizations) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Validation Services Market Overview
• Pharmaceutical Validation Services market size has reached to $25.43 billion in 2025 • Expected to grow to $35.96 billion in 2030 at a compound annual growth rate (CAGR) of 7.2% • Growth Driver: Growth In BioPharmaceuticals And Biosimilars Driving The Market Growth Due To Increasing Regulatory Validation Requirements • Market Trend: Next-Generation Validation Platforms Accelerate Growth And Optimize Processes • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmaceutical Validation Services Market?
Pharmaceutical validation services are specialized quality assurance services that verify and document that pharmaceutical facilities, equipment, processes, and systems consistently perform according to predefined specifications and regulatory standards. These services help to assure product quality, patient safety, and regulatory compliance throughout pharmaceutical manufacturing operations. The main services types of pharmaceutical validation service include installation qualification, operational qualification, performance qualification, and revalidation. Installation qualification refers to the documented verification that equipment or systems are installed according to manufacturer specifications and regulatory requirements. These services are provided for product types including active pharmaceutical ingredient (API) validation, finished pharmaceutical product validation, biologics and biosimilars validation, and medical device validation. They are delivered through onsite validation services and remote validation services, and are used by end users including pharmaceutical manufacturers, biopharmaceutical companies, contract development and manufacturing organizations (CDMOs), and contract research organizations (CROs).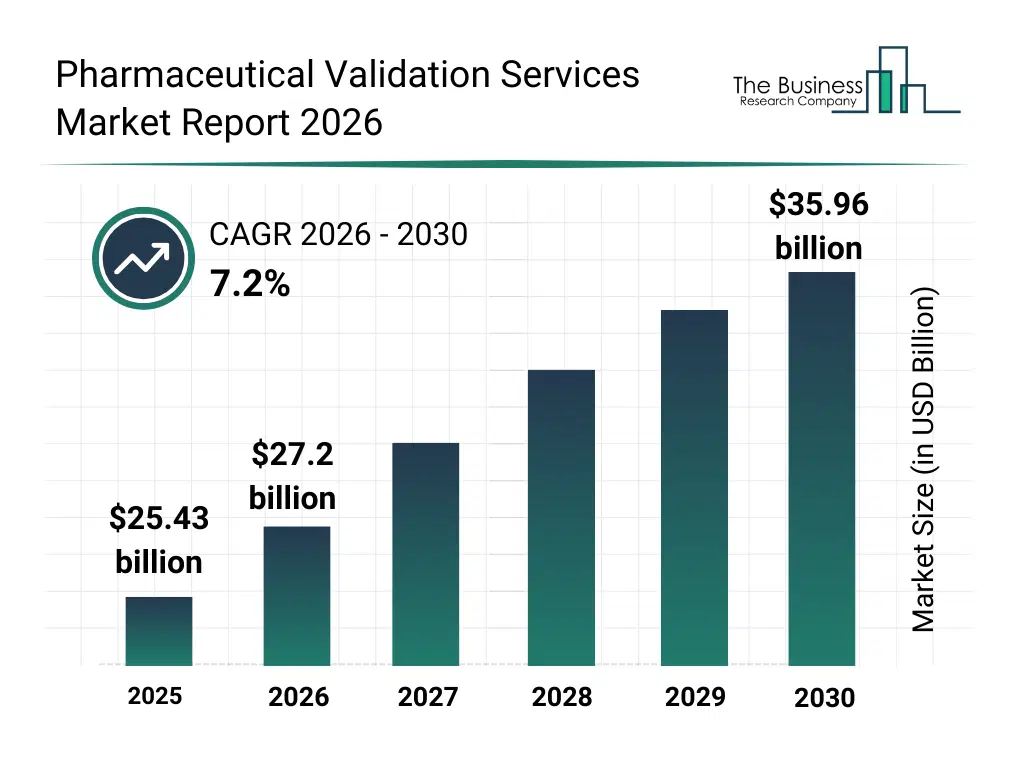
What Is The Pharmaceutical Validation Services Market Size and Share 2026?
The pharmaceutical validation services market size has grown strongly in recent years. It will grow from $25.43 billion in 2025 to $27.2 billion in 2026 at a compound annual growth rate (CAGR) of 7.0%. The growth in the historic period can be attributed to tightening regulatory compliance norms, expansion of gmp facilities, growth in pharma automation, rise in audit observations, increase in validation documentation needs.What Is The Pharmaceutical Validation Services Market Growth Forecast?
The pharmaceutical validation services market size is expected to see strong growth in the next few years. It will grow to $35.96 billion in 2030 at a compound annual growth rate (CAGR) of 7.2%. The growth in the forecast period can be attributed to growth in advanced therapy manufacturing, expansion of continuous manufacturing, higher data integrity enforcement, rising biologics facility builds, demand for lifecycle validation programs. Major trends in the forecast period include digital validation documentation, automated equipment qualification, remote validation audits, integrated process validation suites, data integrity validation platforms.Global Pharmaceutical Validation Services Market Segmentation
1) By Service Type: Installation Qualification; Operational Qualification; Performance Qualification; Revalidation 2) By Product Type: Active Pharmaceutical Ingredient Validation; Finished Pharmaceutical Product Validation; Biologics and Biosimilars Validation; Medical Device Validation 3) By Delivery Mode: Onsite Validation Services; Remote Validation Services 4) By End-User: Pharmaceutical Manufacturers; Biopharmaceutical Companies; Contract Development and Manufacturing Organizations; Contract Research Organizations Subsegments: 1) By Installation Qualification: Equipment Installation Verification; Utility Connection Verification; Component Identification and Documentation; Calibration Verification; Safety Compliance Verification 2) By Operational Qualification: Operational Parameter Testing; Alarm and Control Verification; Standard Operating Procedure Verification; Process Control Testing; Worst Case Condition Testing 3) By Performance Qualification: Process Consistency Verification; Product Quality Performance Testing; Long Term Process Monitoring; Batch Reproducibility Assessment; End Use Performance Validation 4) By Revalidation: Periodic Revalidation; Change Control Based Revalidation; Equipment Modification Revalidation; Process Change Revalidation; Regulatory Compliance RevalidationWhat Is The Driver Of The Pharmaceutical Validation Services Market?
The growth in biopharmaceuticals and biosimilars is expected to propel the growth of the pharmaceutical validation services market going forward. Biopharmaceuticals and biosimilars are therapeutic products derived from biological sources that require rigorous testing and validation processes to ensure safety and efficacy. The rise of biopharmaceuticals and biosimilars is driven by the increasing prevalence of chronic and complex diseases, which require advanced, targeted therapies that traditional small-molecule drugs cannot effectively address. Pharmaceutical validation services ensure the quality, safety, and efficacy of biopharmaceuticals and biosimilars by systematically verifying that manufacturing processes, equipment, and analytical methods consistently produce products that meet stringent regulatory standards, thereby enabling reliable scale-up, regulatory approval, and patient trust in these complex biologic therapies. For instance, in January 2023, according to Cardinal Health Inc., a US-based healthcare company, as of January 2022, the FDA had approved 33 biosimilar products in the US, of which 21 were commercially available. By 2023, the number of FDA-approved biosimilars had increased to 40, with 25 available on the market. Therefore, growth in biopharmaceuticals and biosimilars is driving the growth of the pharmaceutical validation services industry.Key Players In The Global Pharmaceutical Validation Services Market
Major companies operating in the pharmaceutical validation services market are Jacobs Engineering Group Inc., Parexel International Corporation, Sartorius AG, ECS Group Ltd., Computer Aid Inc., ZenQ Ltd., PQE Group S.p.A., SQA Services Ltd., PharmEng Technology Inc., Lachman Consultants Pvt. Ltd., Quality by Design Group Ltd., BioPharma Services Inc., ValSource Inc., Körber AG, PharmOut Consulting Ltd., VMP Consulting Pvt. Ltd., GxP Compliance Ltd., Pharmaligent LLC, Farbridge Pharma Consulting Ltd., and Adris Pharma Validation & Compliance Pvt. Ltd.Global Pharmaceutical Validation Services Market Trends and Insights
Major companies operating in the pharmaceutical validation services market are focusing on developing innovative solutions, such as artificial intelligence (AI)‑enabled digital validation platforms, to streamline compliance, reduce manual errors, and accelerate product approval cycles. AI‑enabled digital validation platforms are software systems that use artificial intelligence to automate and integrate validation processes, helping reduce manual errors, ensure regulatory compliance, and speed up documentation and approval cycles. For instance, in June 2025, ValGenesis Inc., a US–based digital validation lifecycle management company, launched ValGenesis Smart GxP, an innovative AI-enabled platform designed to unify validation and process development across the product lifecycle, integrating multiple applications such as iVal, iClean, and iOps to automate commissioning, qualification, cleaning validation, and real-time operational data capture, with its AI assistant delivering up to 80% faster document generation and reducing review cycles from weeks to hours while ensuring compliance with global regulatory standards. ValGenesis Smart GxP supports seamless data flow from R&D through commercial manufacturing, enhances visibility and traceability, and accelerates time to market, representing a significant advancement over legacy systems in efficiency, compliance, and digital integration.What Are Latest Mergers And Acquisitions In The Pharmaceutical Validation Services Market?
In December 2024, QbD Group NV, a Belgium-based life sciences support company, acquired SciencePharma Sp. z o.o. for an undisclosed amount. With this acquisition, QbD Group NV aims to expand its international footprint and enhance its regulatory and product development capabilities, particularly in Eastern Europe and China. SciencePharma Sp. z o.o. is a Poland-based healthcare consultancy that specializes in regulatory affairs, CMC services, and product development support for the pharmaceutical industry.Regional Insights
North America was the largest region in the pharmaceutical validation services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Validation Services Market?
The pharmaceutical validation services market includes revenues earned by entities through the process validation, cleaning validation, equipment qualification, facility and utility validation, computer system validation, analytical method validation, data integrity assessment, risk management, and documentation support. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Validation Services Market Report 2026?
The pharmaceutical validation services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical validation services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Validation Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $27.2 billion |
| Revenue Forecast In 2035 | $35.96 billion |
| Growth Rate | CAGR of 7.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Product Type, Delivery Mode, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Jacobs Engineering Group Inc., Parexel International Corporation, Sartorius AG, ECS Group Ltd., Computer Aid Inc., ZenQ Ltd., PQE Group S.p.A., SQA Services Ltd., PharmEng Technology Inc., Lachman Consultants Pvt. Ltd., Quality by Design Group Ltd., BioPharma Services Inc., ValSource Inc., Körber AG, PharmOut Consulting Ltd., VMP Consulting Pvt. Ltd., GxP Compliance Ltd., Pharmaligent LLC, Farbridge Pharma Consulting Ltd., and Adris Pharma Validation & Compliance Pvt. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Pharmaceutical Validation Services market was valued at $25.43 billion in 2025, increased to $27.2 billion in 2026, and is projected to reach $35.96 billion by 2030.
request a sample hereThe global Pharmaceutical Validation Services market is expected to grow at a CAGR of 7.2% from 2026 to 2035 to reach $35.96 billion by 2035.
request a sample hereSome Key Players in the Pharmaceutical Validation Services market Include, Jacobs Engineering Group Inc., Parexel International Corporation, Sartorius AG, ECS Group Ltd., Computer Aid Inc., ZenQ Ltd., PQE Group S.p.A., SQA Services Ltd., PharmEng Technology Inc., Lachman Consultants Pvt. Ltd., Quality by Design Group Ltd., BioPharma Services Inc., ValSource Inc., Körber AG, PharmOut Consulting Ltd., VMP Consulting Pvt. Ltd., GxP Compliance Ltd., Pharmaligent LLC, Farbridge Pharma Consulting Ltd., and Adris Pharma Validation & Compliance Pvt. Ltd. .
request a sample hereMajor trend in this market includes: Next-Generation Validation Platforms Accelerate Growth And Optimize Processes. For further insights on this market.
request a sample hereNorth America was the largest region in the pharmaceutical validation services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the pharmaceutical validation services market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here