Pharmaceutical Analytical Testing Outsourcing Market Report 2026

Pharmaceutical Analytical Testing Outsourcing Market Report 2026
Global Outlook – By Service Type (Method Development and Validation, Stability Testing, Microbial Testing, Raw Material Testing, Other Service Types), By Product Type (Small Molecules and Generics, Biologics and Biosimilars, Cell and Gene Therapies, Vaccines, Medical Devices), By Application (Clinical, Preclinical, Other Applications), By End-User (Pharmaceutical Companies, Biotechnology Companies, Contract Research Organizations, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Analytical Testing Outsourcing Market Overview
• Pharmaceutical Analytical Testing Outsourcing market size has reached to $9.43 billion in 2025 • Expected to grow to $14.6 billion in 2030 at a compound annual growth rate (CAGR) of 9.2% • Growth Driver: Rising Complexity Of Drug Molecules Fueling The Growth Of The Market Due To Increasing Development Of Biologics, Peptides, And Advanced Drug Formulations • Market Trend: Advancements In Automation And Label-Free Phenotypic Screening Accelerating Drug Discovery Efficiency • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmaceutical Analytical Testing Outsourcing Market?
Pharmaceutical analytical testing outsourcing refers to the practice of contracting third-party laboratories or service providers to perform analytical testing of pharmaceutical products. helps pharmaceutical companies access specialized expertise, advanced equipment, and scalable testing capacity without heavy in-house investment. It helps to ensure accurate, reliable, and regulatory-compliant testing of drugs while reducing costs and operational burden for pharmaceutical manufacturers. The main services types of pharmaceutical analytical testing outsourcing include method development and validation, stability testing, microbial testing, raw material testing, and other service types. Method development and validation refers to the design and optimization of analytical procedures to ensure accuracy, precision, and compliance with regulatory standards. These services are applied for product types including small molecules and generics, biologics and biosimilars, cell and gene therapies, vaccines, and medical devices. They are used across applications such as clinical, preclinical, and other applications, and are utilized by end users including pharmaceutical companies, biotechnology companies, contract research organizations (CROs), and other end users.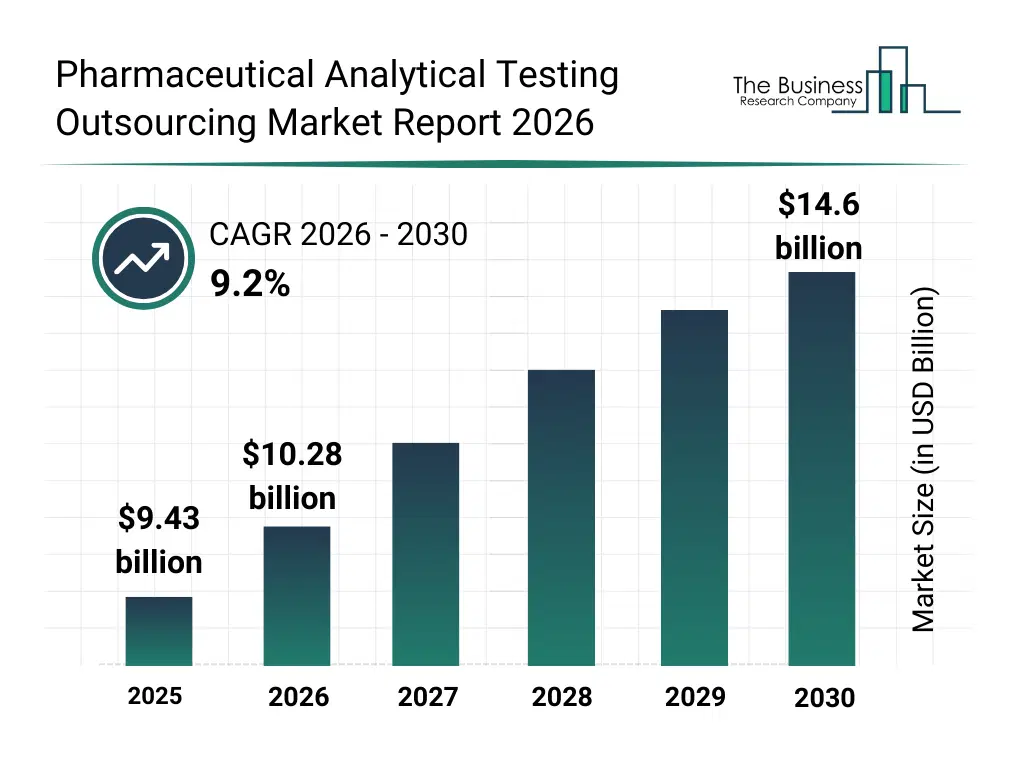
What Is The Pharmaceutical Analytical Testing Outsourcing Market Size and Share 2026?
The pharmaceutical analytical testing outsourcing market size has grown strongly in recent years. It will grow from $9.43 billion in 2025 to $10.28 billion in 2026 at a compound annual growth rate (CAGR) of 8.9%. The growth in the historic period can be attributed to rising drug development complexity, high in house lab costs, growth in contract research organizations, stricter regulatory testing needs, expansion of generic drug pipelines.What Is The Pharmaceutical Analytical Testing Outsourcing Market Growth Forecast?
The pharmaceutical analytical testing outsourcing market size is expected to see strong growth in the next few years. It will grow to $14.6 billion in 2030 at a compound annual growth rate (CAGR) of 9.2%. The growth in the forecast period can be attributed to growth in biologics and cell therapies, increasing regulatory scrutiny, expansion of virtual pharma models, rising small biotech outsourcing, demand for faster batch release. Major trends in the forecast period include specialized third party analytical labs, integrated method validation services, high throughput bioanalytical testing, regulatory ready testing packages, end to end quality control outsourcing.Global Pharmaceutical Analytical Testing Outsourcing Market Segmentation
1) By Service Type: Method Development and Validation; Stability Testing; Microbial Testing; Raw Material Testing; Other Service Types 2) By Product Type: Small Molecules and Generics; Biologics and Biosimilars; Cell and Gene Therapies; Vaccines; Medical Devices 3) By Application: Clinical; Preclinical; Other Applications 4) By End-User: Pharmaceutical Companies; Biotechnology Companies; Contract Research Organizations; Other End-Users Subsegments: 1) By Method Development and Validation: Analytical Method Development; Analytical Method Validation; Method Transfer Studies; Assay Optimization; Robustness Testing 2) By Stability Testing: Accelerated Stability Testing; Long Term Stability Testing; Intermediate Stability Testing; Photostability Testing; Stress Stability Testing 3) By Microbial Testing: Bioburden Testing; Sterility Testing; Endotoxin Testing; Microbial Limits Testing; Environmental Monitoring 4) By Raw Material Testing: Identity Testing; Purity Testing; Physicochemical Characterization; Impurity Profiling; Supplier Qualification Testing 5) By Other Service Types: Process Development Support; Regulatory Documentation Support; Quality Control Testing; Batch Release Testing; Technical ConsultingWhat Is The Driver Of The Pharmaceutical Analytical Testing Outsourcing Market?
The rising complexity of drug molecules is expected to propel the growth of the pharmaceutical analytical testing outsourcing market. Drug molecule complexity refers to the increasing structural, chemical, and functional sophistication of modern therapeutics, including biologics, peptides, oligonucleotides, and targeted therapies that require highly precise characterization and validation. This complexity is being driven by advancements in specialty and precision medicine, which demand advanced analytical techniques to ensure product quality, safety, and regulatory compliance. As in-house analytical capabilities often struggle to meet the technical and regulatory demands of complex molecules, pharmaceutical companies increasingly rely on specialized third-party analytical testing providers. Outsourcing provides access to advanced instrumentation, expert knowledge, and scalable capacity that are often impractical or too costly to maintain internally. For instance, according to MedChem Australia, an Australia-based national drug discovery initiative, around 55 new drugs were approved in 2023, including 17 biologics and 9 peptide- or oligonucleotide-based therapies (TIDES). These modalities require sophisticated analytical methods such as mass spectrometry, bioassays, and advanced stability testing, increasing reliance on outsourced analytical services. Therefore, the rising complexity of drug molecules is driving the growth of the pharmaceutical analytical testing outsourcing industry.Key Players In The Global Pharmaceutical Analytical Testing Outsourcing Market
Major companies operating in the pharmaceutical analytical testing outsourcing market are Thermo Fisher Scientific Inc., IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON plc, SGS Société Générale de Surveillance SA, Eurofins Scientific SE, WuXi AppTec Co. Ltd., Intertek Group plc, Charles River Laboratories International Inc., West Pharmaceutical Services Inc., Element Materials Technology Group Limited, Recipharm AB, Almac Group Limited, Pace Analytical Services LLC, Cotecna Inspection S.A., BioAgilytix Labs LLC, BA Sciences Inc., Toxikon Corporation, Frontage Laboratories Inc., Syngene International LimitedGlobal Pharmaceutical Analytical Testing Outsourcing Market Trends and Insights
Major companies operating in the pharmaceutical analytical testing outsourcing market are focusing on advancing automated and digital analytical testing platforms to improve the accuracy, efficiency, and regulatory compliance of quality control (QC) testing across the drug development and manufacturing lifecycle. These advancements aim to enhance method robustness, increase throughput, and ensure reliable impurity profiling, identity confirmation, and batch release testing in accordance with regulatory requirements. For instance, in March 2023, Eurofins Discovery, a US-based contract research organization (CRO), expanded its analytical testing services by introducing automated, label-free mass spectrometry–based analytical platforms to support regulated pharmaceutical quality control applications. These platforms enable high-throughput, reproducible analysis of drug substances and drug products without reliance on fluorescent or radioactive labels, preserving sample integrity and improving data reliability. The enhanced capabilities support critical QC activities such as impurity detection, identity testing, and method validation, enabling pharmaceutical manufacturers to meet stringent regulatory standards while accelerating testing timelines.What Are Latest Mergers And Acquisitions In The Pharmaceutical Analytical Testing Outsourcing Market?
In December 2024, Novo Holdings A/S, a Denmark-based private holding and investment company, acquired Catalent, Inc., for an undisclosed amount. Through this acquisition, Novo Holdings aims to strengthen its life sciences investment portfolio by supporting Catalent’s expansion as a leading global contract development and manufacturing organization, enhancing capabilities across pharmaceutical development, manufacturing, analytical testing, and full life-cycle supply for pharma and biotech companies. Catalent, Inc., is a US-based contract development and manufacturing organization (CDMO) that provides outsourced services to pharmaceutical, biotechnology, and consumer health companies across the full product lifecycle.Regional Insights
North America was the largest region in the pharmaceutical analytical testing outsourcing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Analytical Testing Outsourcing Market?
The pharmaceutical analytical testing outsourcing market includes revenues earned by entities through the analytical method development, analytical method validation, bioanalytical testing services, impurity profiling, contract research services, quality control testing, formulation analysis, bioequivalence studies, and regulatory support services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pharmaceutical Analytical Testing Outsourcing Market Report 2026?
The pharmaceutical analytical testing outsourcing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical analytical testing outsourcing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Analytical Testing Outsourcing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $10.28 billion |
| Revenue Forecast In 2035 | $14.6 billion |
| Growth Rate | CAGR of 8.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Product Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON plc, SGS Société Générale de Surveillance SA, Eurofins Scientific SE, WuXi AppTec Co. Ltd., Intertek Group plc, Charles River Laboratories International Inc., West Pharmaceutical Services Inc., Element Materials Technology Group Limited, Recipharm AB, Almac Group Limited, Pace Analytical Services LLC, Cotecna Inspection S.A., BioAgilytix Labs LLC, BA Sciences Inc., Toxikon Corporation, Frontage Laboratories Inc., Syngene International Limited |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Pharmaceutical Analytical Testing Outsourcing market was valued at $9.43 billion in 2025, increased to $10.28 billion in 2026, and is projected to reach $14.6 billion by 2030.
request a sample hereThe global Pharmaceutical Analytical Testing Outsourcing market is expected to grow at a CAGR of 9.2% from 2026 to 2035 to reach $14.6 billion by 2035.
request a sample hereSome Key Players in the Pharmaceutical Analytical Testing Outsourcing market Include, Thermo Fisher Scientific Inc., IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON plc, SGS Société Générale de Surveillance SA, Eurofins Scientific SE, WuXi AppTec Co. Ltd., Intertek Group plc, Charles River Laboratories International Inc., West Pharmaceutical Services Inc., Element Materials Technology Group Limited, Recipharm AB, Almac Group Limited, Pace Analytical Services LLC, Cotecna Inspection S.A., BioAgilytix Labs LLC, BA Sciences Inc., Toxikon Corporation, Frontage Laboratories Inc., Syngene International Limited .
request a sample hereMajor trend in this market includes: Advancements In Automation And Label-Free Phenotypic Screening Accelerating Drug Discovery Efficiency. For further insights on this market.
request a sample hereNorth America was the largest region in the pharmaceutical analytical testing outsourcing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the pharmaceutical analytical testing outsourcing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here