Chronic Idiopathic Urticaria Treatment Market Report 2026

Chronic Idiopathic Urticaria Treatment Market Report 2026
Global Outlook – By Types (Tablets, Capsules, Creams, Lotions, Injections), By Drug Class (Antihistamines, Corticosteroids, Leukotriene Receptor Antagonists, Immunosuppressants, Biologic Therapies, Other Drug Class), By Route Of Administration (Oral, Injectable, Topical), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies) – Market Size, Trends, Strategies, and Forecast to 2035
Chronic Idiopathic Urticaria Treatment Market Overview
• Chronic Idiopathic Urticaria Treatment market size has reached to $7.24 billion in 2025 • Expected to grow to $11.23 billion in 2030 at a compound annual growth rate (CAGR) of 9.2% • Growth Driver: The Increasing Focus On Biologics Is Fueling Market Growth Due To High Demand For Targeted Therapies • Market Trend: Innovative Targeted Treatments For Chronic Urticaria Offering Effective Alternatives To Traditional Therapy • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Chronic Idiopathic Urticaria Treatment Market?
Chronic idiopathic urticaria treatment refers to the medical management strategies used to control long-lasting hives of unknown cause, aiming to reduce itching, swelling, and flare-ups while improving patient quality of life through symptom suppression and prevention of recurrences. It typically involves a stepwise approach using non-sedating antihistamines as first-line therapy, followed by dose escalation, add-on biologic therapies, immunosuppressants, or short courses of corticosteroids for patients with moderate to severe or treatment-resistant symptoms. The main types of chronic idiopathic urticaria treatment include tablets, capsules, creams, lotions, and injections. Tablets refer to oral solid formulations used to relieve symptoms of chronic urticaria. These treatments belong to drug classes such as antihistamines, corticosteroids, leukotriene receptor antagonists, immunosuppressants, biologic therapies, and other drug classes. They are administered and distributed through channels including oral, injectable, and topical routes via hospital pharmacies, retail pharmacies, and online pharmacies.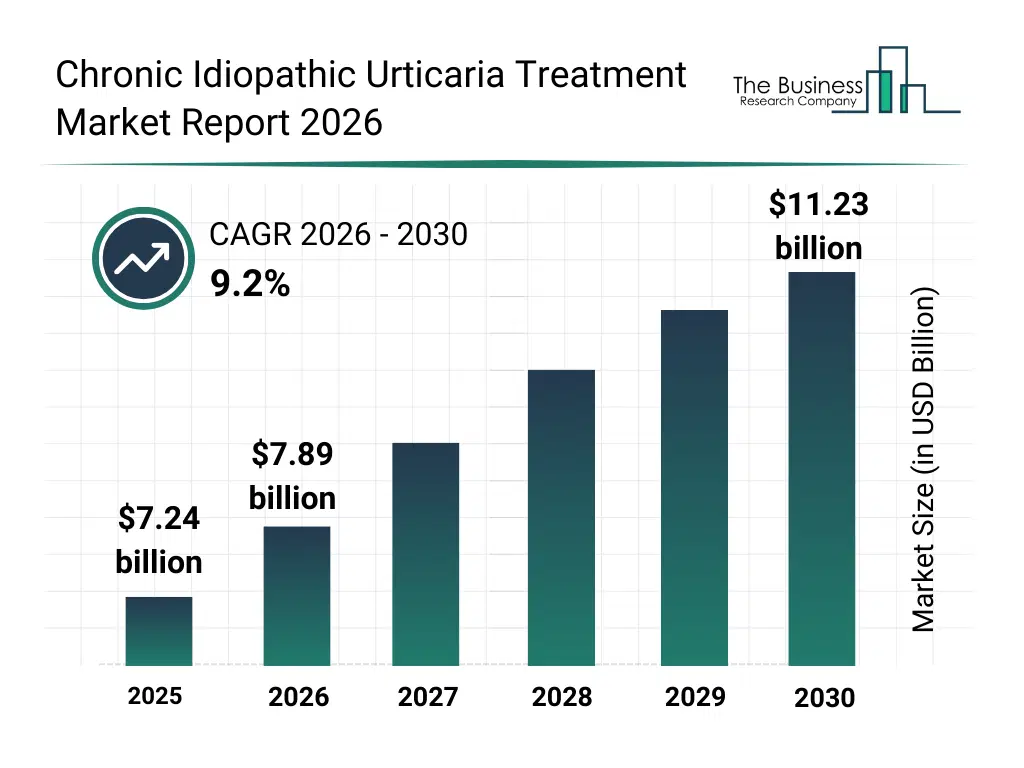
What Is The Chronic Idiopathic Urticaria Treatment Market Size and Share 2026?
The chronic idiopathic urticaria treatment market size has grown strongly in recent years. It will grow from $7.24 billion in 2025 to $7.89 billion in 2026 at a compound annual growth rate (CAGR) of 9.0%. The growth in the historic period can be attributed to increasing diagnosis of chronic urticaria, availability of second-line antihistamine therapies, adoption of stepwise treatment protocols, expansion of allergy specialty clinics, growing use of immunomodulatory drugs.What Is The Chronic Idiopathic Urticaria Treatment Market Growth Forecast?
The chronic idiopathic urticaria treatment market size is expected to see strong growth in the next few years. It will grow to $11.23 billion in 2030 at a compound annual growth rate (CAGR) of 9.2%. The growth in the forecast period can be attributed to increasing demand for biologic treatment options, rising focus on personalized immunology care, expansion of digital disease management tools, growing investment in novel urticaria therapies, increasing patient awareness and treatment adherence. Major trends in the forecast period include increasing adoption of biologic therapies for refractory patients, growing use of personalized treatment pathways, rising integration of digital symptom monitoring tools, expansion of injectable treatment options, enhanced focus on long-term disease control.Global Chronic Idiopathic Urticaria Treatment Market Segmentation
1) By Types: Tablets; Capsules; Creams; Lotions; Injections 2) By Drug Class: Antihistamines; Corticosteroids; Leukotriene Receptor Antagonists; Immunosuppressants; Biologic Therapies; Other Drug Class 3) By Route Of Administration: Oral; Injectable; Topical 4) By Distribution Channel: Hospital Pharmacies; Retail Pharmacies; Online Pharmacies Subsegments: 1) By Tablets: Immediate Release Tablets; Extended Release Tablets; Enteric Coated Tablets; Chewable Tablets; Orally Disintegrating Tablets 2) By Capsules: Hard Gelatin Capsules; Soft Gelatin Capsules; Sustained Release Capsules; Delayed Release Capsules 3) By Creams: Medicated Topical Creams; Anti Inflammatory Creams; Antifungal Creams; Antibacterial Creams; Moisturizing Therapeutic Creams 4) By Lotions: Medicated Therapeutic Lotions; Anti Inflammatory Lotions; Antiseptic Lotions; Moisturizing Lotions; Specialty Dermatological Lotions 5) By Injections: Intravenous Injections; Intramuscular Injections; Subcutaneous Injections; Pre Filled Syringe Injections; Auto Injector Based InjectionsWhat Is The Driver Of The Chronic Idiopathic Urticaria Treatment Market?
The increasing focus on biologics is expected to propel the growth of the chronic idiopathic urticaria treatment market going forward. Biologics are therapeutic products derived from living organisms or their components that target specific pathways in the immune system to treat diseases. The emphasis on biologics is growing due to advancements in biotechnology and immunology, which have enabled the development of more precise and effective treatments that target the underlying mechanisms of immune-mediated conditions rather than merely managing symptoms. Biologics target the underlying immune pathways and mast cell activation responsible for chronic idiopathic urticaria, revolutionizing treatment paradigms and providing patients with more effective and long-lasting symptom control. For instance, in February 2025, according to the World Health Organization (WHO), a Switzerland-based intergovernmental organization, the 2023 WHO Essential Medicines List includes 81 biologic therapies, representing over 15% of all listed medicines. Therefore, the increasing focus on biologics is driving the growth of chronic idiopathic urticaria treatment industry.Key Players In The Global Chronic Idiopathic Urticaria Treatment Market
Major companies operating in the chronic idiopathic urticaria treatment market are F. Hoffmann-La Roche Ltd., Sanofi S.A., Novartis AG., AstraZeneca PLC, GlaxoSmithKline plc, Teva Pharmaceutical Industries Ltd., Regeneron Pharmaceuticals Inc., Incyte Corporation, Dr. Reddy’s Laboratories Ltd., Celltrion Inc., Yuhan Corporation, Kiniksa Pharmaceuticals Ltd., Celldex Therapeutics Inc., Septerna Therapeutics Inc., Carna Biosciences Inc., United BioPharma Inc., Recludix Pharma Inc., Jasper Therapeutics Inc., Evommune Inc., ARS Pharmaceuticals IncGlobal Chronic Idiopathic Urticaria Treatment Market Trends and Insights
Major companies operating in the chronic idiopathic urticaria treatment market are focusing on developing innovative products such as monoclonal antibody therapies to provide targeted, long-lasting relief for patients unresponsive to standard treatments. Monoclonal antibody therapies are biologic drugs designed to specifically target immune system components, such as immunoglobulin E (IgE) or other inflammatory mediators, that trigger chronic hives, offering more precise action compared to traditional antihistamines or corticosteroids. For instance, in September 2025, Celltrion Inc., a South Korea-based biopharmaceutical company, launched Omriclo, an omalizumab biosimilar, in Norway. The product is composed of the same active monoclonal antibody as Xolair, functions by neutralizing circulating IgE, and is administered via subcutaneous injection. Its unique features include first-mover advantage in Europe, enhanced accessibility through local distribution channels, and potential cost savings for healthcare systems. It is intended for patients with chronic idiopathic urticaria who remain symptomatic despite standard therapy, providing a biologic alternative to conventional treatments and offering improved symptom management, reduced frequency of flare-ups, and the potential to enhance overall quality of life by minimizing the reliance on long-term antihistamine or corticosteroid use.What Are Latest Mergers And Acquisitions In The Chronic Idiopathic Urticaria Treatment Market?
In March 2025, Novartis AG, a Switzerland-based pharmaceutical company, acquired worldwide rights to KRP-M223, a preclinical drug from Kyorin Pharmaceutical Co. Ltd., for an upfront payment of $55 million. With this acquisition, Novartis AG aims to develop, produce, and commercialize KRP-M223, an MRGPRX2 antagonist designed to treat chronic spontaneous urticaria (CSU) and other mast cell-driven inflammatory conditions, while Kyorin retains the option to manufacture and market the drug within Japan. Kyorin Pharmaceutical Co. Ltd. is a Japan-based pharmaceutical company specializing in providing chronic idiopathic urticaria treatment.Regional Insights
North America was the largest region in the chronic idiopathic urticaria treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Chronic Idiopathic Urticaria Treatment Market?
The chronic idiopathic urticaria treatment market includes revenues earned by entities through clinical diagnosis and disease assessment, allergy and trigger evaluation services, personalized treatment planning, antihistamine therapy management, and treatment response assessments. The market value includes the value of related digital tools and devices sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Chronic Idiopathic Urticaria Treatment Market Report 2026?
The chronic idiopathic urticaria treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the chronic idiopathic urticaria treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Chronic Idiopathic Urticaria Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $7.89 billion |
| Revenue Forecast In 2035 | $11.23 billion |
| Growth Rate | CAGR of 9.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Types, Drug Class, Route Of Administration, Distribution Channel |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Sanofi S.A., Novartis AG., AstraZeneca PLC, GlaxoSmithKline plc, Teva Pharmaceutical Industries Ltd., Regeneron Pharmaceuticals Inc., Incyte Corporation, Dr. Reddy’s Laboratories Ltd., Celltrion Inc., Yuhan Corporation, Kiniksa Pharmaceuticals Ltd., Celldex Therapeutics Inc., Septerna Therapeutics Inc., Carna Biosciences Inc., United BioPharma Inc., Recludix Pharma Inc., Jasper Therapeutics Inc., Evommune Inc., ARS Pharmaceuticals Inc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Chronic Idiopathic Urticaria Treatment market was valued at $7.24 billion in 2025, increased to $7.89 billion in 2026, and is projected to reach $11.23 billion by 2030.
request a sample hereThe global Chronic Idiopathic Urticaria Treatment market is expected to grow at a CAGR of 9.2% from 2026 to 2035 to reach $11.23 billion by 2035.
request a sample hereSome Key Players in the Chronic Idiopathic Urticaria Treatment market Include, F. Hoffmann-La Roche Ltd., Sanofi S.A., Novartis AG., AstraZeneca PLC, GlaxoSmithKline plc, Teva Pharmaceutical Industries Ltd., Regeneron Pharmaceuticals Inc., Incyte Corporation, Dr. Reddy’s Laboratories Ltd., Celltrion Inc., Yuhan Corporation, Kiniksa Pharmaceuticals Ltd., Celldex Therapeutics Inc., Septerna Therapeutics Inc., Carna Biosciences Inc., United BioPharma Inc., Recludix Pharma Inc., Jasper Therapeutics Inc., Evommune Inc., ARS Pharmaceuticals Inc .
request a sample hereMajor trend in this market includes: Innovative Targeted Treatments For Chronic Urticaria Offering Effective Alternatives To Traditional Therapy. For further insights on this market.
request a sample hereNorth America was the largest region in the chronic idiopathic urticaria treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the chronic idiopathic urticaria treatment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here