Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market Report 2026

Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market Report 2026
Global Outlook – By Product Type (Assay Kits, Instruments, Reagents And Consumables), By Application (Cervical Cancer Screening, Research, Other Applications), By End User (Hospitals And Clinics, Diagnostic Laboratories, Research Institutes, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market Overview
• Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing market size has reached to $1.06 billion in 2025 • Expected to grow to $2.12 billion in 2030 at a compound annual growth rate (CAGR) of 14.9% • Growth Driver: Increasing Prevalence Of Cervical Cancer Fueling The Growth Of The Market Due To Rising Disease Burden And Need For Early Molecular Detection • Market Trend: Innovations in Multiplex Real-Time PCR Technology Enhance HPV E6/E7 mRNA Detection Specificity • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market?
Cervical human papillomavirus (HPV) messenger ribonucleic acid (mRNA) testing refers to a molecular diagnostic method that detects the expression of E6/E7 oncogenic mRNA from high-risk HPV types in cervical samples, indicating active viral infection that is biologically involved in cervical cell transformation. Unlike HPV DNA tests that identify the presence of viral genetic material, mRNA testing is more specific for clinically significant infections associated with a higher risk of cervical precancer and cancer. The main product types of cervical human papillomavirus (HPV) messenger ribonucleic acid (mRNA) testing include assay kits, instruments, and reagents and consumables. Assay kits refer to ready-to-use kits designed to detect and quantify HPV mRNA for the early diagnosis and screening of cervical cancer. These products are applied across areas including cervical cancer screening, research, and other applications, and are used by end users including hospitals and clinics, diagnostic laboratories, research institutes, and other end users.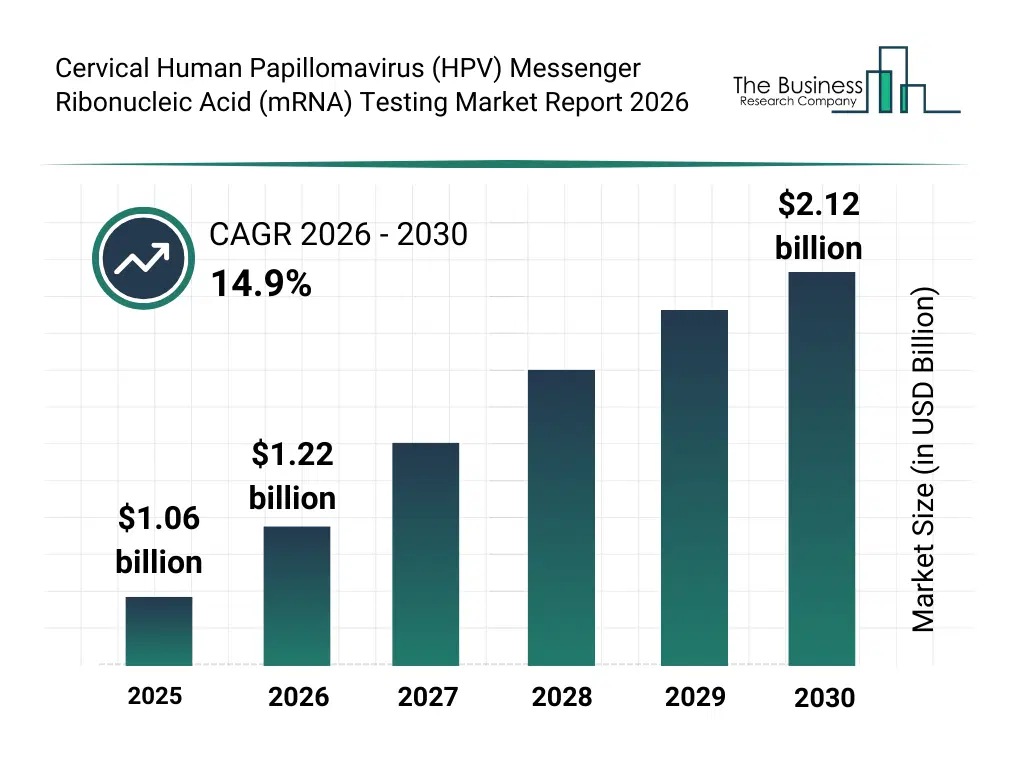
What Is The Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market Size and Share 2026?
The cervical human papillomavirus (hpv) messenger ribonucleic acid (mrna) testing market size has grown rapidly in recent years. It will grow from $1.06 billion in 2025 to $1.22 billion in 2026 at a compound annual growth rate (CAGR) of 14.6%. The growth in the historic period can be attributed to increasing cervical cancer screening program coverage, adoption of molecular diagnostics in gynecology, rising awareness of hr-hpv infection risks, availability of automated pcr platforms, expansion of centralized diagnostic laboratories.What Is The Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market Growth Forecast?
The cervical human papillomavirus (hpv) messenger ribonucleic acid (mrna) testing market size is expected to see rapid growth in the next few years. It will grow to $2.12 billion in 2030 at a compound annual growth rate (CAGR) of 14.9%. The growth in the forecast period can be attributed to increasing shift toward primary hpv screening, growing demand for high-specificity cancer risk stratification, expansion of population-based screening programs, integration of digital pathology and reporting systems, increasing investments in precision diagnostics. Major trends in the forecast period include increasing adoption of mrna-based primary cervical screening assays, growing use of fully automated molecular testing platforms, rising integration of genotype-specific reflex testing, expansion of high-specificity screening algorithms, enhanced focus on clinically actionable hpv detection.Global Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market Segmentation
1) By Product Type: Assay Kits; Instruments; Reagents And Consumables 2) By Application: Cervical Cancer Screening; Research; Other Applications 3) By End User: Hospitals And Clinics; Diagnostic Laboratories; Research Institutes; Other End Users Subsegments: 1) By Assay Kits: Messenger Ribonucleic Acid Detection Kits; Multiplex Testing Kits; High Risk Genotype Specific Kits; Screening And Diagnostic Kits 2) By Instruments: Real Time Polymerase Chain Reaction Systems; Automated Molecular Analyzers; Sample Preparation Systems; Nucleic Acid Extraction Instruments 3) By Reagents And Consumables: Primers And Probes; Enzymes And Buffers; Controls And Standards; Extraction And Purification ConsumablesWhat Is The Driver Of The Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market?
The increasing prevalence of cervical cancer are expected to propel the growth of the cervical human papillomavirus messenger ribonucleic acid testing market going forward. Cervical cancer is a malignant disease that develops in the cells of the cervix, primarily caused by persistent infection with high-risk human papillomavirus types, and it represents a significant cause of cancer-related illness and death among women worldwide. The increasing prevalence of cervical cancer is increasing due to low cervical screening participation, leading to delayed detection and progression of persistent human papillomavirus infections into cancer. Cervical human papillomavirus messenger ribonucleic acid testing supports increasing prevalence of cervical cancer identification of active high-risk human papillomavirus infections, improving risk stratification, and supporting timely intervention to reduce cervical cancer progression. For instance, in December 2025, according to Cancer Australia, an Australia-based a Government Agency, In 2023 there were 265 deaths from cervical cancer in australia in 2025, it is estimated that there will be 254 deaths from cervical cancer. Therefore, the increasing prevalence of cervical cancer is driving the growth of the cervical human papillomavirus messenger ribonucleic acid testing market.Key Players In The Global Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market
Major companies operating in the cervical human papillomavirus (hpv) messenger ribonucleic acid (mrna) testing market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Becton, Dickinson and Company, Eurofins Scientific, Agilent Technologies Inc., bioMérieux SA, Bio-Rad Laboratories Inc., Bio-Techne Corporation, Co-Dx Ltd., Promega Corporation, Seegene Inc., Molbio Diagnostics, QIAGEN N.V., Sansure Biotech Inc., Xiamen Zeesan Biotech Co. Ltd., DiaCarta Inc., Operon S.A., AB ANALITICA s.r.l., Arbor Vita Corporation, Mylab Discovery Solutions, Guangdong Hybribio Biotech Co. Ltd., Atila Biosystems, GeneFirst Ltd., Advanced Molecular Diagnostics.Global Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market Trends and Insights
Major companies operating in the cervical human papillomavirus messenger ribonucleic acid (mRNA) testing market are focusing on developing technological advancements, such as multiplex real-time PCR innovations, to gain competitive advantage by improving diagnostic accuracy, enabling simultaneous E6/E7 mRNA detection and high-risk HPV genotyping, reducing false positives, lowering testing costs, and expanding adoption. Multiplex real-time PCR innovations refers to the ability to detect and quantify multiple HPV E6/E7 mRNA targets and high-risk genotypes simultaneously in a single PCR reaction, improving accuracy, efficiency, and clinical relevance of cervical HPV testing. For instance, July 2024, Medicon Hellas S.A., a Greece based diagnostics company, introduced the multiplex real time pcr kit for hpv e6/e7 mrna genotyping as the exclusive distributor for uni-medica in greece and cyprus. The kit features compatibility with multiple pcr instruments like bio-rad cfx96 and hongshi slan-96 series for fluorochrome analysis with ct values, applicability to diverse samples including semen, urine, and anal swabs beyond cytology, and ce-ivd marking with results in 2.5 hours.What Are Latest Mergers And Acquisitions In The Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market?
In April 2025, Institut Pasteur, a France-based provider of biomedical research, translational science, and infectious disease innovation, partnered with ABL Diagnostics. With this partnership, the strategic objective is to commercialize and scale an advanced hpv rna sequencing technology by integrating institut pasteur’s hpv rna-seq intellectual property into abl diagnostics molecular diagnostic portfolio, thereby strengthening technological capabilities and expanding offerings in high-risk hpv molecular diagnostics. ABL Diagnostics is a France-based provider of molecular diagnostic solutions specializing in viral load testing, infectious disease assays, and advanced RNA-based diagnostic technologies for clinical and public health applications.Regional Insights
North America was the largest region in the cervical human papillomavirus (HPV) messenger ribonucleic acid (mRNA) testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market?
The cervical human papillomavirus (HPV) messenger ribonucleic acid (mRNA) testing market consists of sales of molecular diagnostic products, primary cervical screening assays, genotyping add-ons, fully automated platform assays, laboratory-developed tests, experimental kits, and integrated screening panels. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market Report 2026?
The cervical human papillomavirus (hpv) messenger ribonucleic acid (mrna) testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the cervical human papillomavirus (hpv) messenger ribonucleic acid (mrna) testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.22 billion |
| Revenue Forecast In 2035 | $2.12 billion |
| Growth Rate | CAGR of 14.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Becton, Dickinson and Company, Eurofins Scientific, Agilent Technologies Inc., bioMérieux SA, Bio-Rad Laboratories Inc., Bio-Techne Corporation, Co-Dx Ltd., Promega Corporation, Seegene Inc., Molbio Diagnostics, QIAGEN N.V., Sansure Biotech Inc., Xiamen Zeesan Biotech Co. Ltd., DiaCarta Inc., Operon S.A., AB ANALITICA s.r.l., Arbor Vita Corporation, Mylab Discovery Solutions, Guangdong Hybribio Biotech Co. Ltd., Atila Biosystems, GeneFirst Ltd., Advanced Molecular Diagnostics. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing market was valued at $1.06 billion in 2025, increased to $1.22 billion in 2026, and is projected to reach $2.12 billion by 2030.
request a sample hereThe global Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing market is expected to grow at a CAGR of 14.9% from 2026 to 2035 to reach $2.12 billion by 2035.
request a sample hereSome Key Players in the Cervical Human Papillomavirus (HPV) Messenger Ribonucleic Acid (mRNA) Testing market Include, F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Becton, Dickinson and Company, Eurofins Scientific, Agilent Technologies Inc., bioMérieux SA, Bio-Rad Laboratories Inc., Bio-Techne Corporation, Co-Dx Ltd., Promega Corporation, Seegene Inc., Molbio Diagnostics, QIAGEN N.V., Sansure Biotech Inc., Xiamen Zeesan Biotech Co. Ltd., DiaCarta Inc., Operon S.A., AB ANALITICA s.r.l., Arbor Vita Corporation, Mylab Discovery Solutions, Guangdong Hybribio Biotech Co. Ltd., Atila Biosystems, GeneFirst Ltd., Advanced Molecular Diagnostics. .
request a sample hereMajor trend in this market includes: Innovations in Multiplex Real-Time PCR Technology Enhance HPV E6/E7 mRNA Detection Specificity. For further insights on this market.
request a sample hereNorth America was the largest region in the cervical human papillomavirus (HPV) messenger ribonucleic acid (mRNA) testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the cervical human papillomavirus (hpv) messenger ribonucleic acid (mrna) testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here