Biologics Active Pharmaceutical Ingredient (API) Market Report 2026

Biologics Active Pharmaceutical Ingredient (API) Market Report 2026
Global Outlook – By Type Of Modality (Monoclonal Antibodies, Recombinant Proteins, Vaccines, Enzymes, Other Biologics), By Scale Of Operation (Preclinical Or Clinical Scale, Commercial Scale), By Service (Upstream Processing, Downstream Purification, Formulation And Fill-Finish Services, Analytical Testing And Quality Control), By End-Users (Pharmaceutical Companies, Biotechnology Companies, Academic And Research Institutions, Contract Research Organizations) – Market Size, Trends, Strategies, and Forecast to 2035
Biologics Active Pharmaceutical Ingredient (API) Market Overview
• Biologics Active Pharmaceutical Ingredient (API) market size has reached to $66.82 billion in 2025 • Expected to grow to $101.84 billion in 2030 at a compound annual growth rate (CAGR) of 8.8% • Growth Driver: Surge In Demand For Personalized Medicine Fueling The Growth Of The Market Due To Increasing Adoption Of Targeted And Biomarker-driven Therapies • Market Trend: Innovations In Biosimilar Development Platforms Enhance Accessibility And Cost Efficiency In Biologics Active Pharmaceutical Ingredients (APIs) • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Biologics Active Pharmaceutical Ingredient (API) Market?
Biologics active pharmaceutical ingredient (API) is a biologically derived substance used as the active component in biopharmaceutical products to diagnose, treat, or prevent diseases. It is produced using living systems such as microorganisms, cell cultures, or recombinant deoxyribonucleic acid technology, and may include monoclonal antibodies, recombinant proteins, vaccines, enzymes, and other complex biologics. Biologics active pharmaceutical ingredient (API) are integral to the efficacy, safety, and quality of biologic drugs and are critical in drug discovery, development, and commercialization. By providing consistent, high-quality biologics active pharmaceutical ingredient (API), manufacturers enable pharmaceutical companies to develop advanced therapies, improve patient outcomes, and accelerate time-to-market. The main types of biologics active pharmaceutical ingredient (API) include monoclonal antibodies, recombinant proteins, vaccines, enzymes, and other biologics. Monoclonal antibodies refer to laboratory-produced molecules engineered to serve as substitute antibodies that can restore, enhance, or mimic the immune system’s attack on target cells. These APIs are produced at scales such as preclinical or clinical scale and commercial scale. They are available in services including upstream processing, downstream purification, formulation and fill-finish services, and analytical testing and quality control, and are used by end users including pharmaceutical companies, biotechnology companies, academic and research institutions, and contract research organizations.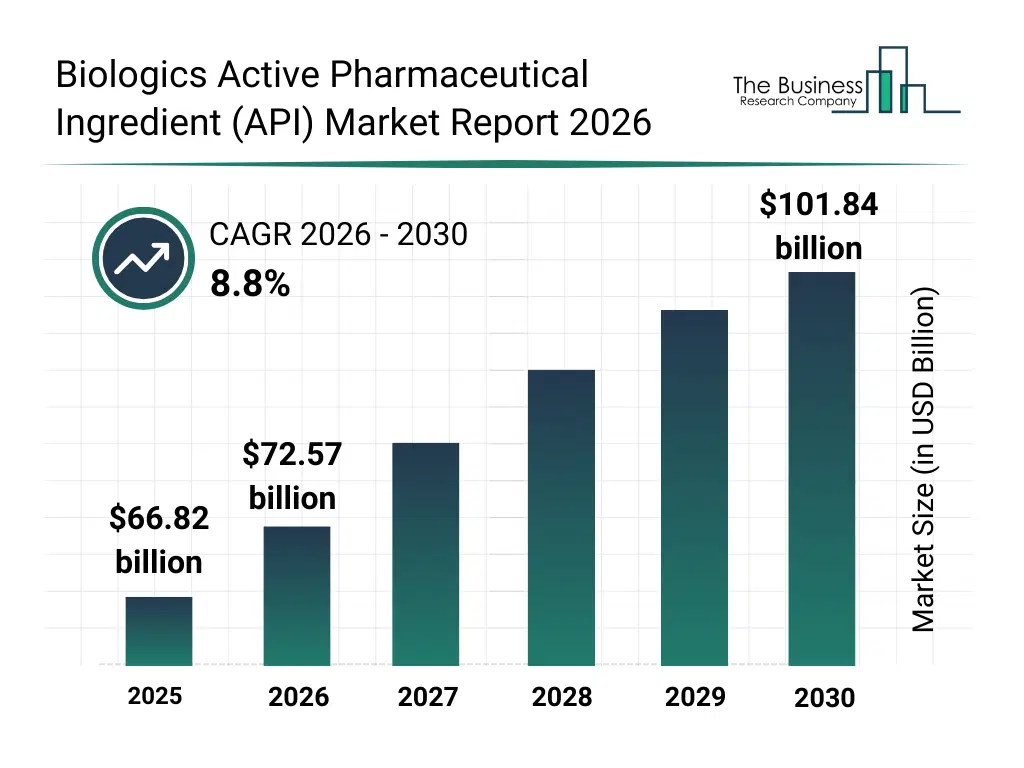
What Is The Biologics Active Pharmaceutical Ingredient (API) Market Size and Share 2026?
The biologics active pharmaceutical ingredient (api) market size has grown strongly in recent years. It will grow from $66.82 billion in 2025 to $72.57 billion in 2026 at a compound annual growth rate (CAGR) of 8.6%. The growth in the historic period can be attributed to increasing prevalence of chronic and autoimmune diseases, expansion of monoclonal antibody development pipelines, growth of recombinant protein therapeutics, rising investments in biologics manufacturing infrastructure, increasing outsourcing to biologics cmos.What Is The Biologics Active Pharmaceutical Ingredient (API) Market Growth Forecast?
The biologics active pharmaceutical ingredient (api) market size is expected to see strong growth in the next few years. It will grow to $101.84 billion in 2030 at a compound annual growth rate (CAGR) of 8.8%. The growth in the forecast period can be attributed to increasing development of cell and gene therapies, rising demand for personalized biologic medicines, expansion of commercial-scale biologics production, growing focus on process scalability and consistency, increasing regulatory emphasis on api quality and traceability. Major trends in the forecast period include increasing adoption of single-use bioprocessing systems, rising demand for high-purity biologics apis, expansion of large-scale commercial biomanufacturing, growing focus on cold chain integrity, enhanced integration of advanced purification technologies.Global Biologics Active Pharmaceutical Ingredient (API) Market Segmentation
1) By Type Of Modality: Monoclonal Antibodies; Recombinant Proteins; Vaccines; Enzymes; Other Biologics 2) By Scale Of Operation: Preclinical Or Clinical Scale; Commercial Scale 3) By Service: Upstream Processing; Downstream Purification; Formulation And Fill-Finish Services; Analytical Testing And Quality Control 4) By End-Users: Pharmaceutical Companies; Biotechnology Companies; Academic And Research Institutions; Contract Research Organizations Subsegments: 1) By Monoclonal Antibodies: Therapeutic Monoclonal Antibodies; Diagnostic Monoclonal Antibodies; Human Monoclonal Antibodies 2) By Recombinant Proteins: Hormonal Proteins; Growth Factor Proteins; Cytokine Proteins 3) By Vaccines: Live Attenuated Vaccines; Inactivated Vaccines; Subunit Vaccines 4) By Enzymes: Replacement Enzymes; Digestive Enzymes; Metabolic Enzymes 5) By Other Biologics: Cell And Gene Therapy Products; Fusion Proteins; Plasma Derived ProductsWhat Is The Driver Of The Biologics Active Pharmaceutical Ingredient (API) Market?
The rising demand for personalized medicine is expected to propel the growth of the biologics active pharmaceutical ingredients market going forward. Personalized medicine refers to a medical approach that tailors disease prevention, diagnosis, and treatment strategies to an individual’s genetic profile, biomarkers, lifestyle, and environmental factors. The rising demand for personalized medicine is driven by improved treatment effectiveness, as therapies tailored to an individual’s genetic and molecular profile enhance clinical outcomes while reducing adverse drug reactions. Rising demand for personalized medicine supports growth in biologics active pharmaceutical ingredient (API) production by increasing the need for precise, patient-specific biologic compounds, which drives higher demand for complex, high-quality biologic APIs used in precision treatment approaches. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based nonprofit industry alliance, the Food and Drug Administration approved 16 new personalized treatments for rare diseases in 2023, a sharp increase from 6 approvals in 2022. Therefore, the rising demand for personalized medicine is driving the growth of the biologics active pharmaceutical ingredients market.Key Players In The Global Biologics Active Pharmaceutical Ingredient (API) Market
Major companies operating in the biologics active pharmaceutical ingredient (api) market are F Hoffmann-La Roche Ltd, Pfizer Inc, AbbVie Inc, Sanofi SA, Novartis AG, AstraZeneca PLC, Bristol-Myers Squibb Company, Thermo Fisher Scientific Inc, GSK plc, Eli Lilly and Company, Amgen Inc, Danaher Corporation, Boehringer Ingelheim International GmbH, AGC Inc, Biogen Inc, Lonza Group AG, Novo Holdings AS, FUJIFILM Biotechnologies Inc, Curia Global Inc, Almac Group Ltd, Syngene International Limited, and Hepalink USA Inc.Global Biologics Active Pharmaceutical Ingredient (API) Market Trends and Insights
Major companies operating in the biologics active pharmaceutical ingredients (APIs) market are focusing on developing advanced solutions, such as biosimilar development platforms, to enhance drug efficacy and improve treatment accessibility. Biosimilar development platforms refer to frameworks and processes that enable the creation of biologic drugs highly similar to approved reference products, ensuring comparable safety, potency, and quality while addressing affordability and supply challenges. For instance, in March 2023, Enzene Biosciences, an India-based biopharmaceutical company, introduced its biosimilar version of adalimumab, a widely used therapeutic monoclonal antibody. This solution focuses on delivering comparable clinical performance to the originator product while providing a cost-effective option for patients and healthcare systems. The platform uses advanced bioprocessing and analytical techniques to maintain consistent batch quality, achieve high purity, and meet regulatory standards, enabling large-scale manufacturing and a reliable supply. It is engineered to meet the growing demand for accessible biologic therapies across chronic and autoimmune diseases, enabling broader patient reach and sustainable treatment options.What Are Latest Mergers And Acquisitions In The Biologics Active Pharmaceutical Ingredient (API) Market?
In March 2025, HAS Healthcare Advanced Synthesis SA, a Switzerland-based pharmaceutical company, acquired Cerbios-Pharma SA for an undisclosed amount. Through this acquisition, HAS expands its CDMO capabilities to include biologics and hybrid drug substances, enabling end-to-end development and manufacturing of complex APIs such as recombinant proteins and antibody‑drug conjugates. Cerbios-Pharma SA is a Switzerland-based company specializing in the development and manufacturing of both chemical and biological APIs.Regional Insights
North America was the largest region in the biologics active pharmaceutical ingredients (API) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Biologics Active Pharmaceutical Ingredient (API) Market?
The biologics active pharmaceutical ingredient (API) market includes sales of cell culture media, bioreactors, purification systems, filtration devices, storage vials, and cold chain logistics solutions. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Biologics Active Pharmaceutical Ingredient (API) Market Report 2026?
The biologics active pharmaceutical ingredient (api) market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the biologics active pharmaceutical ingredient (api) industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Biologics Active Pharmaceutical Ingredient (API) Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $72.57 billion |
| Revenue Forecast In 2035 | $101.84 billion |
| Growth Rate | CAGR of 8.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type Of Modality, Scale Of Operation, Service, End-Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F Hoffmann-La Roche Ltd, Pfizer Inc, AbbVie Inc, Sanofi SA, Novartis AG, AstraZeneca PLC, Bristol-Myers Squibb Company, Thermo Fisher Scientific Inc, GSK plc, Eli Lilly and Company, Amgen Inc, Danaher Corporation, Boehringer Ingelheim International GmbH, AGC Inc, Biogen Inc, Lonza Group AG, Novo Holdings AS, FUJIFILM Biotechnologies Inc, Curia Global Inc, Almac Group Ltd, Syngene International Limited, and Hepalink USA Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Biologics Active Pharmaceutical Ingredient (API) market was valued at $66.82 billion in 2025, increased to $72.57 billion in 2026, and is projected to reach $101.84 billion by 2030.
request a sample hereThe global Biologics Active Pharmaceutical Ingredient (API) market is expected to grow at a CAGR of 8.8% from 2026 to 2035 to reach $101.84 billion by 2035.
request a sample hereSome Key Players in the Biologics Active Pharmaceutical Ingredient (API) market Include, F Hoffmann-La Roche Ltd, Pfizer Inc, AbbVie Inc, Sanofi SA, Novartis AG, AstraZeneca PLC, Bristol-Myers Squibb Company, Thermo Fisher Scientific Inc, GSK plc, Eli Lilly and Company, Amgen Inc, Danaher Corporation, Boehringer Ingelheim International GmbH, AGC Inc, Biogen Inc, Lonza Group AG, Novo Holdings AS, FUJIFILM Biotechnologies Inc, Curia Global Inc, Almac Group Ltd, Syngene International Limited, and Hepalink USA Inc. .
request a sample hereMajor trend in this market includes: Innovations In Biosimilar Development Platforms Enhance Accessibility And Cost Efficiency In Biologics Active Pharmaceutical Ingredients (APIs). For further insights on this market.
request a sample hereNorth America was the largest region in the biologics active pharmaceutical ingredients (API) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the biologics active pharmaceutical ingredient (api) market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here